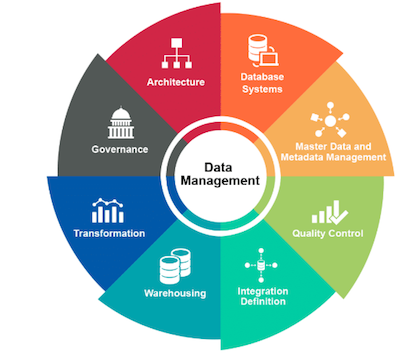
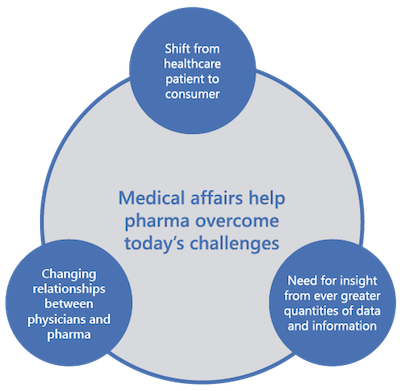
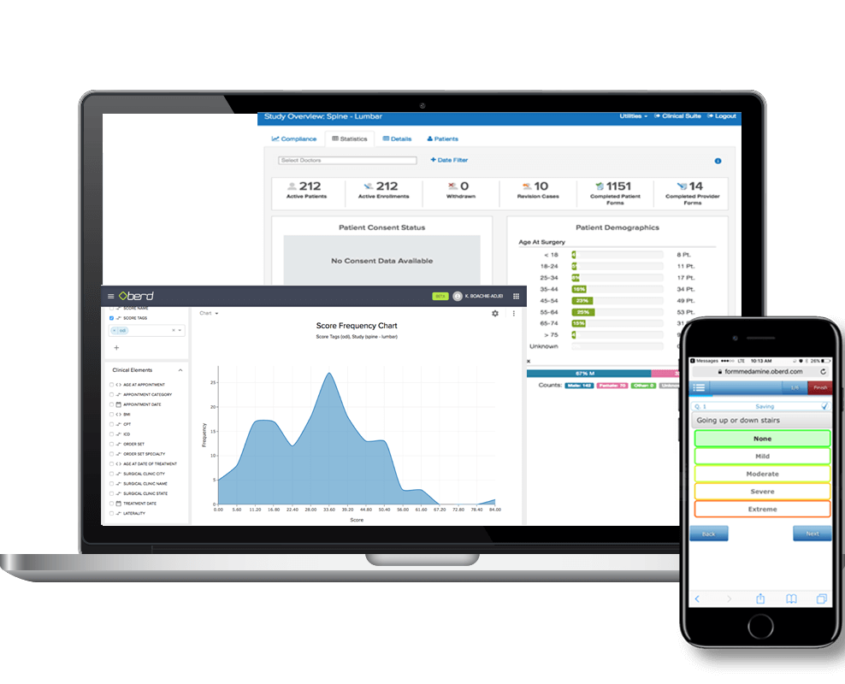
We provide clinical trial related services as a turnkey contract research organization (CRO) to physicians, group private practices, pharmaceutical companies, biotech companies, medical device companies, and clinical sites.
Hospitals and health care institutions that also do not have adequate infrastructure and staff to meet the requirements of clinical trial protocols of interest make up the majority of these clinical sites.
It is our role to be a trusted advisor making decisions and acting with our clients’ best interests in mind. Full services included but not limited to:
| • Study Start-up Services | • Drafting appropriate informed consent forms (ICF) | • Advising and alerting investigators of potential protocol violations | • Clinical Monitoring |
| • Contract negotiations on behalf of the physician with the study sponsor or vice versa | • Site initiation and trial close-out operations | • Data Management | • Clinical Project Management |
| • Protocol submissions for Institutional Review | • Trial-related documents archival and maintenance (CTMS) | • Biostatistics | • Regulatory Affairs and Submissions |
| • Board or Independent Ethics Committee (IRB/IEC) approval | • Reporting serious adverse events to the Sponsor and the IRB | • Pharmacovigilance and Reporting | • IRB Submissions |
| • Patient Follow-up | • Patient Recruitment | • Ensuring protocol compliance | • QA Auditing |
| • Feasibility analysis and site selection for multi-center studies |
Our Services
Clinical Trials
Research Hours
Published Articles